On the Mechanism of Palladium-Catalyzed Aromatic C−H Oxidation | Journal of the American Chemical Society

PDF) Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps

Oxidative Addition of Water, Alcohols, and Amines in Palladium Catalysis - Grünwald - 2020 - Angewandte Chemie International Edition - Wiley Online Library
Elucidating the reaction mechanism of a palladium-palladium dual catalytic process through kinetic studies of proposed elementary steps | Communications Chemistry

Quadruple C-H activation coupled to hydrofunctionalization and C-H silylation/borylation enabled by weakly coordinated palladium catalyst | Nature Communications

Oxidative Addition of Aryl Tosylates to Palladium(0) and Coupling of Unactivated Aryl Tosylates at Room Temperature | Journal of the American Chemical Society

Aerobic Heterogeneous Palladium-Catalyzed Oxidative Allenic C−H Arylation: Benzoquinone as a Direct Redox Mediator between O2 and Pd | CCS Chem

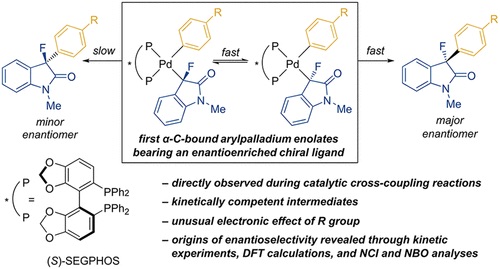
Kinetic Resolution of Benzylamines via Palladium(II)-Catalyzed C–H Cross-Coupling | Journal of the American Chemical Society

Pd-catalyzed asymmetric oxidative C-H/C-H cross-coupling reaction between dialkylaminomethylferrocenes and indolizines - ScienceDirect

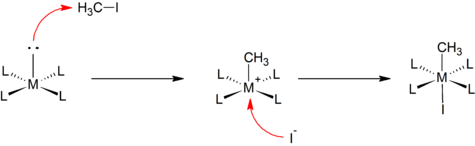
Inorganics | Free Full-Text | Structural Study of Model Rhodium(I) Carbonylation Catalysts Activated by Indole-2-/Indoline-2-Carboxylate Bidentate Ligands and Kinetics of Iodomethane Oxidative Addition

Palladium–Protein Oxidative Addition Complexes by Amine-Selective Acylation | Journal of the American Chemical Society
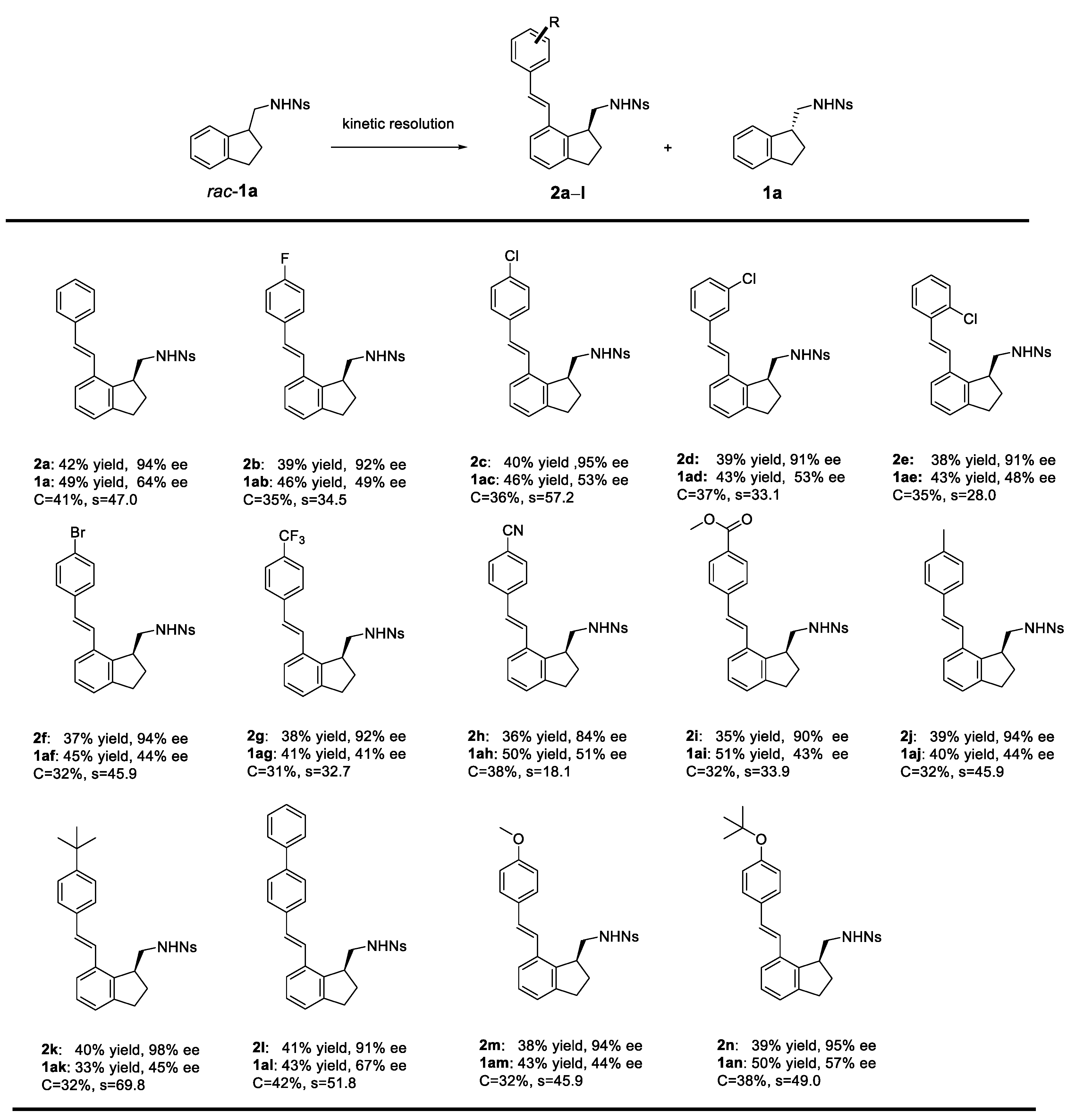
Molecules | Free Full-Text | Kinetic Resolution of β-Alkyl Phenylethylamine Derivatives through Palladium-Catalyzed, Nosylamide-Directed C−H Olefination
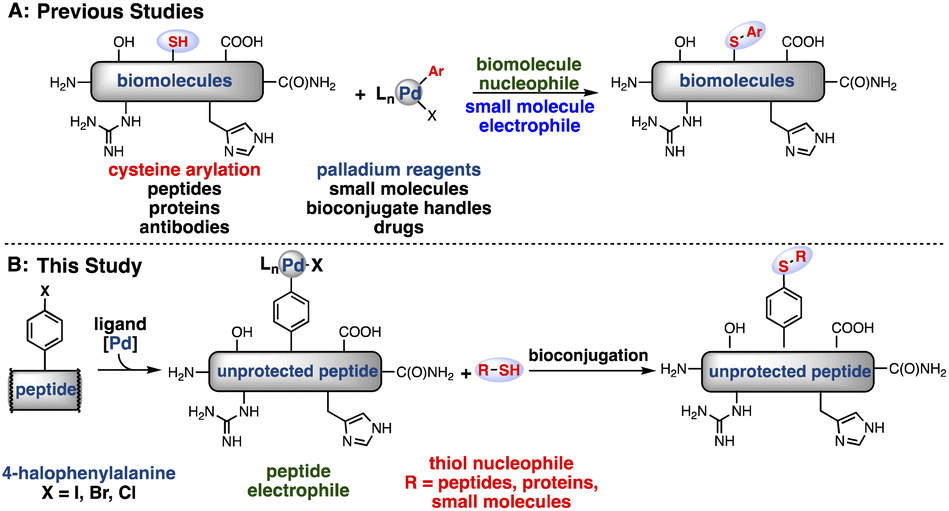
Palladium–peptide oxidative addition complexes for bioconjugation - Chemical Science (RSC Publishing) DOI:10.1039/D2SC04074C

Kinetic Resolution of Allyltriflamides through a Pd-Catalyzed C–H Functionalization with Allenes: Asymmetric Assembly of Tetrahydropyridines | Journal of the American Chemical Society

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -
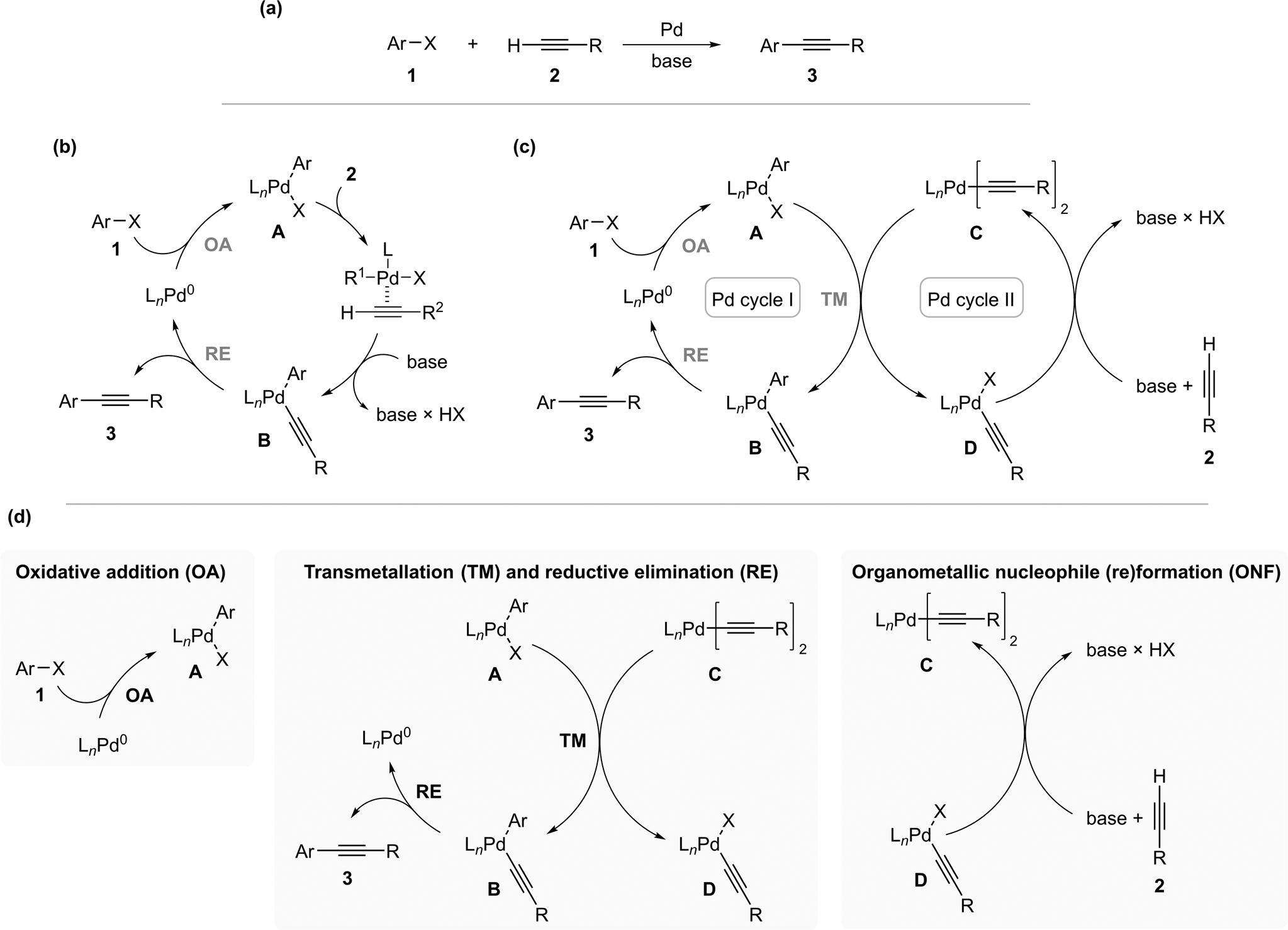
Kinetic Studies of the Oxidative Addition and Transmetallation Steps Involved in the Cross-Coupling of Alkynyl Stannanes with Ar

Dynamic Kinetic Resolution of Biaryl Lactones via a Chiral Bifunctional Amine Thiourea-Catalyzed Highly Atropo-enantioselective Transesterification. | Semantic Scholar
Frontiers | Key Mechanistic Features in Palladium-Catalyzed Methylcyclopropanation of Norbornenes With Vinyl Bromides: Insights From DFT Calculations


![Oxidative addition of aryl halidest o[ Ni(PEt 3 ) 4 ]: (a) Kochi's... | Download Scientific Diagram Oxidative addition of aryl halidest o[ Ni(PEt 3 ) 4 ]: (a) Kochi's... | Download Scientific Diagram](https://www.researchgate.net/publication/320543286/figure/fig3/AS:616378570645530@1523967511774/Oxidative-addition-of-aryl-halidest-o-NiPEt-3-4-a-Kochis-mechanistic-proposal.png)