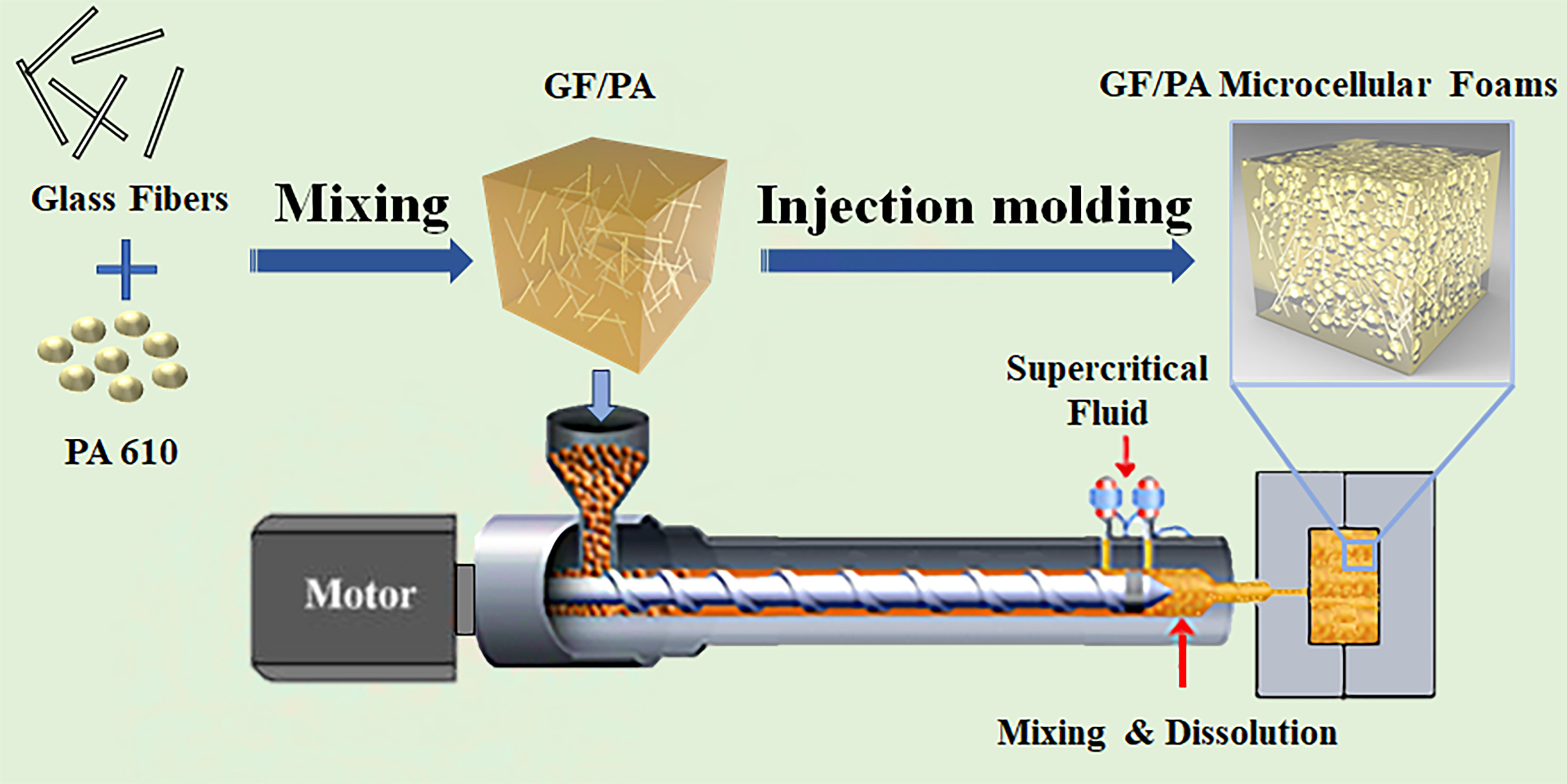
Polymers | Free Full-Text | Microstructure and Properties of Glass Fiber-Reinforced Polyamide/Nylon Microcellular Foamed Composites

So after finally getting a decent print I printed a gear in makerbot nylon with pva support. I put the print in water to dissolve the supports. After removing from the water

SOLVED: Making Nylon 6,10 with Sebacoyl Chloride and 1,6-hexanediamine. 1) Sebacoyl chloride is soluble in hydrocarbons, 1,6-Hexanediamine is soluble in water. How can these facts explain the fact that the reactant solutions

Sensual woman in black nylon stockings sitting on white box isolated on grey - Stock Photo - Dissolve

SEM image of 20% nylon 6/formic acid nanofibers at a voltage of 15 kV... | Download Scientific Diagram

Simple process for separation and recycling of nylon 6 and polyurethane components from waste nylon 6/polyurethane debris - Caihong Gong, Kaihui Zhang, Ce Yang, Juan Chen, Shen Zhang, Chunwang Yi, 2021